
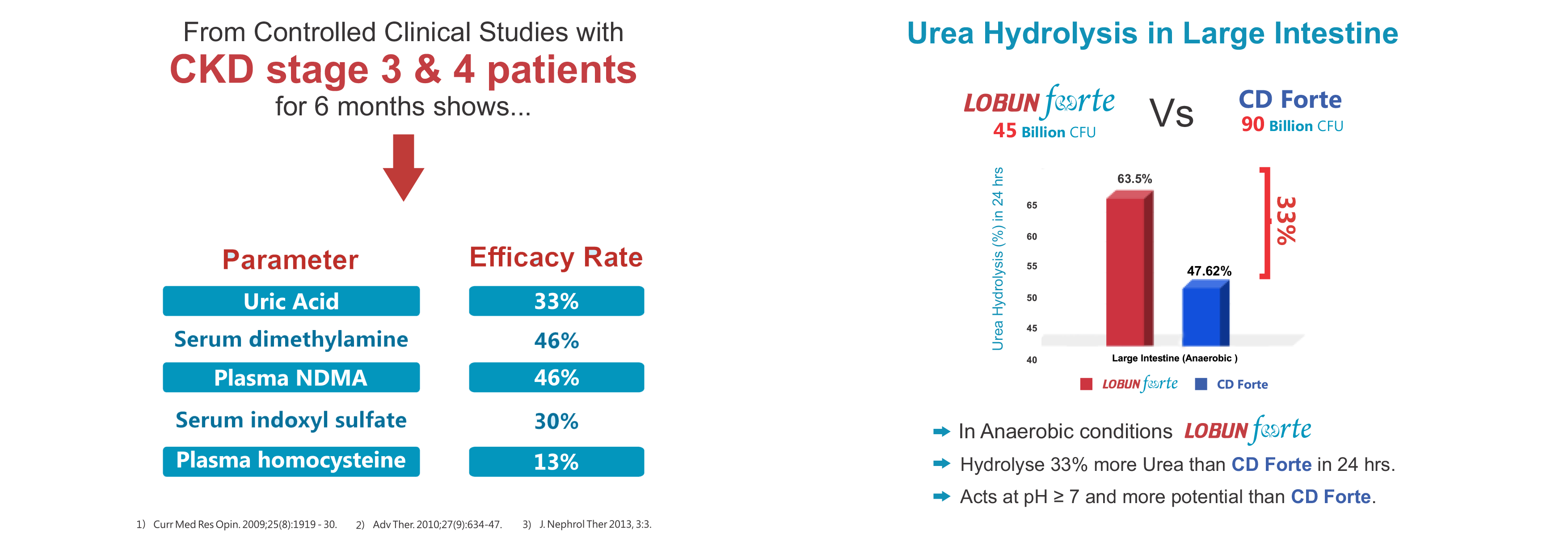
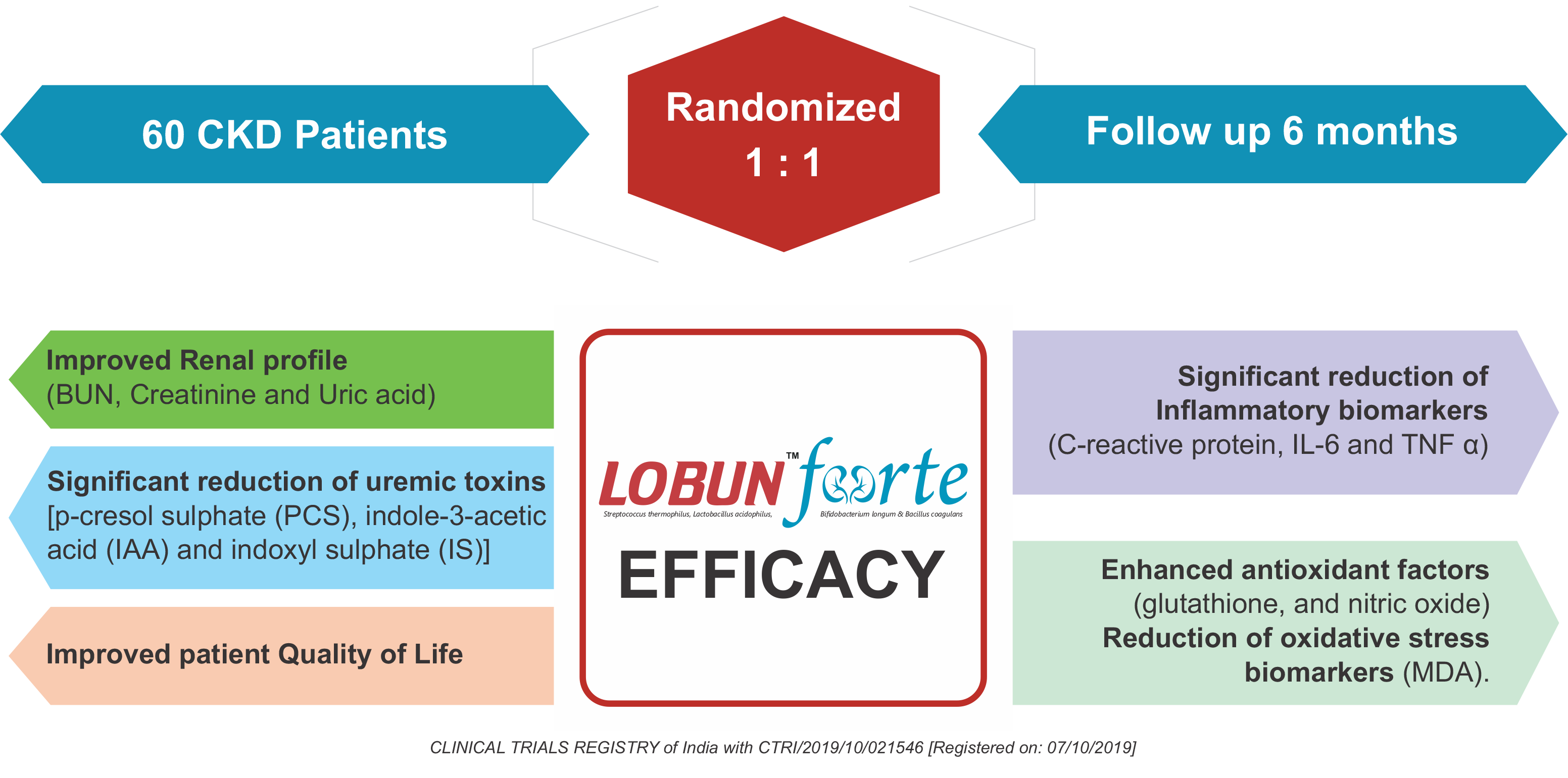
Rapid Reduction of Uremic Toxins in CKD patients

Delay the Progression of CKD from stage 3-4 to End Stage Renal Disease

Formulated by the house of Sanzyme with 50+ yrs experience

Stable shelf life

Unaffected during Gastric transit

Rapid multiplication rate

High urea utilising microbe

After the permeability of gut for passing of toxins

Helps in removal of putrefactive bacteria
Removes p-Cresol and indoxyl toxins
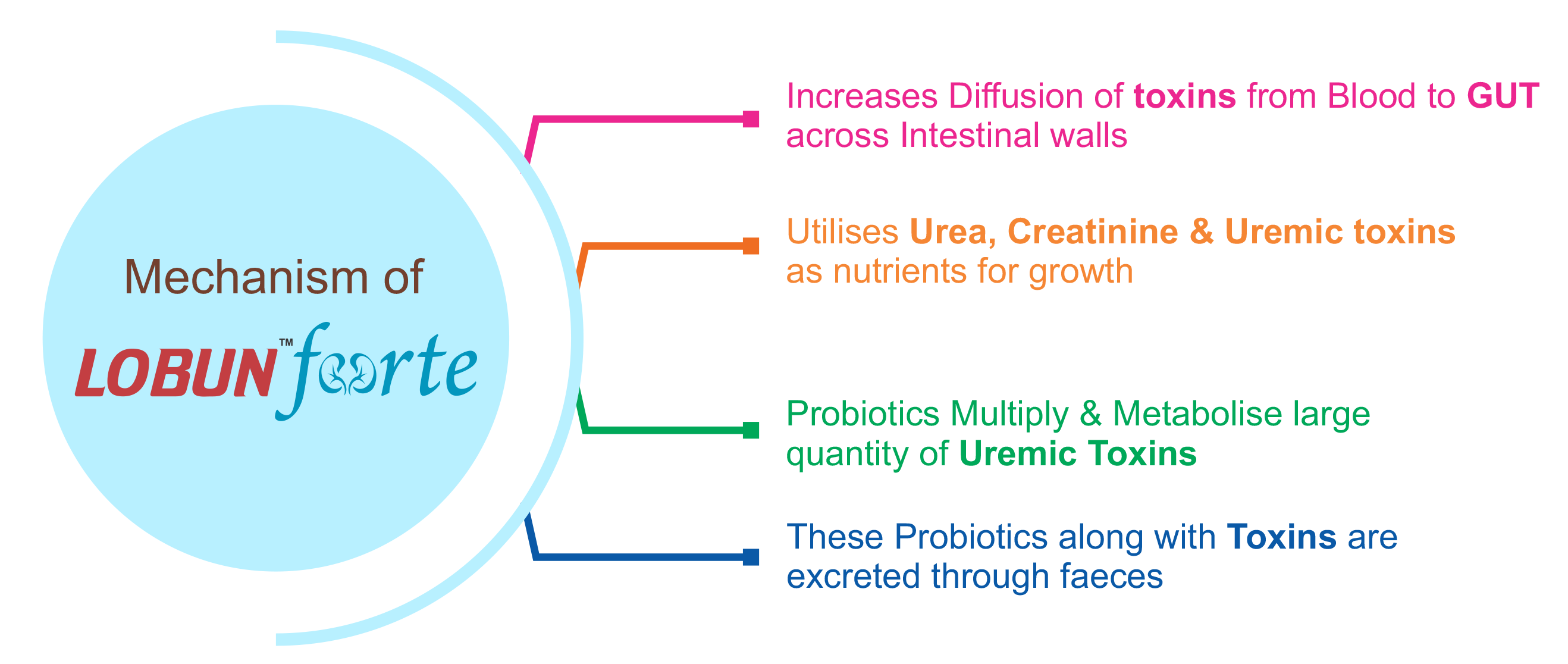
DMA : Dimethyl Amine
NDMA : Nitroso Dimethyl Amine
GFR : Glomerular Filtration Rate
QoL : Quality of Life

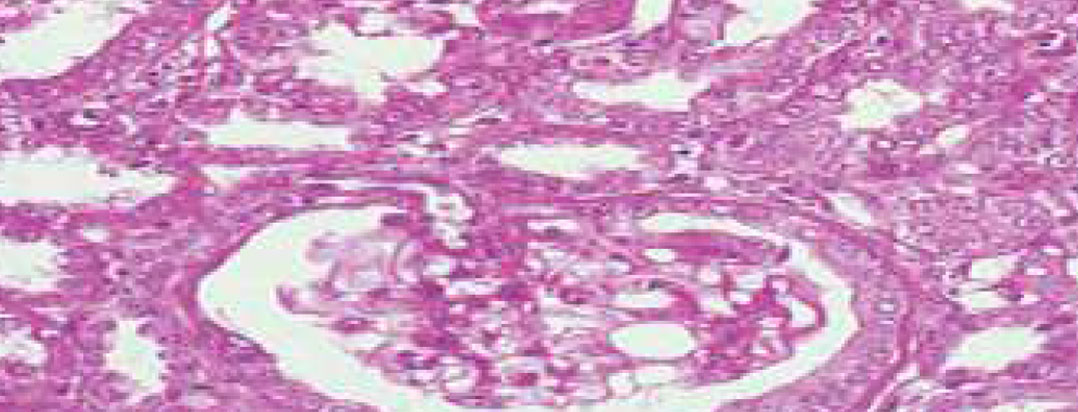
Longitudinal Section of CKD Rat Kidney depicting Renal Tubular Hyperplasia (Cyclosporine induced)
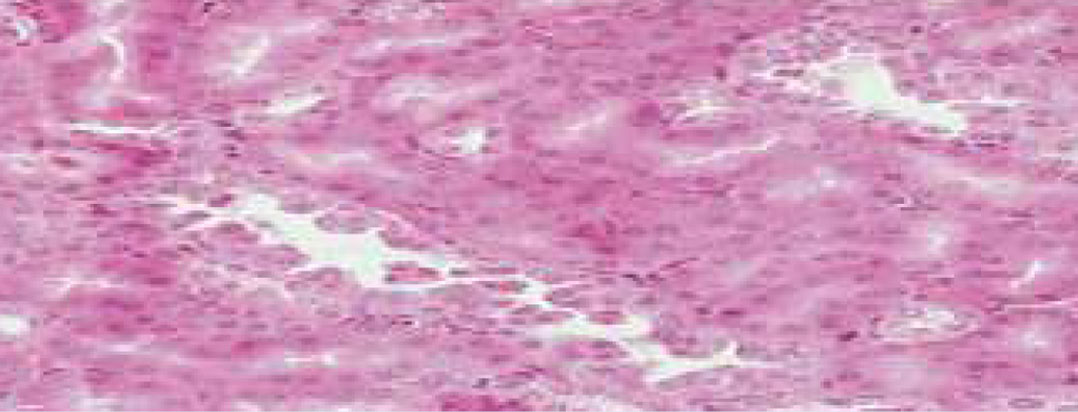
Longitudinal Section of CKD Rat Kidney treated with LOBUN.
| Day 0 | Day 28 | Day 42 | Day 56 | ||
|---|---|---|---|---|---|
| BUN (mg/dL) | Cyclosporine induced CKD rats | 9.75 | 28.5 | 35.0 | 40.5 |
| Cyclosporine induced CKD rats treated with Lobun | 9.95 | 20.75 | 14.87 | 12.4 | |
| Serum Creatinine (mg/dL) | Cyclosporine induced CKD rats | 0.75 | 2.72 | 4.35 | 6.62 |
| Cyclosporine induced CKD rats treated with Lobun | 0.57 | 1.67 | 1.47 | 0.85 |



info@sanzyme.com
Phone : 040-69959999
Phone : 040-48589999
2nd Floor, Sattva Signature Tower,
H.No. 8-2-472/1/A/B/SF-3,
Road No. 1, Banjara Hills,
Hyderabad, Telangana (India) – 500034